Tumor transplantation model - human tumor cell line xenograft CDX and human tumor tissue source xenograft PDX
Q: What is a tumor transplant model? Human-derived tumor cell line xenograft (CDX), which is to inoculate tumor cells subcultured in vitro to immunodeficient mice, usually in the subcutaneous, intravenous or in situ. Human tumor derived xenograft (PDX), a tumor model established by directly transplanting fresh tumor tissue of a patient into immunodeficient mice. The common inoculation site is subcutaneous and in situ. The specific process is as shown below. The response to cetuximab treatment and genetic profile of GC-PDX models. Panel A: The PDX-GC models are sorted by the tumor response to cetuximab (DT/DC). The responders at the right part display higher EGFR mRNA level And IHC staining intensity, and the only two cases (GA0075 and GA0152, CN . 5) of gene amplification. Panel B: The representative images of responders and non-responders. The responders GA0152 and GA0075 display IHC score 31, and gene amplification ( GA0075, CN 5 5.8; GA0152, CN . 15), while non-responders GA0119 and GA0139 are with IHC low Hand-Held Rice Transplanter,Efficient Hand-Held Rice Transplanter,Human Hand-Held Rice Transplanter,Hand-Held Small Rice Transplanter Taizhou Yingtian Agricultural Machinery Manufacturing Co., Ltd. , https://www.sakuradaagc.com
A: The humanized tumor model constructed by transplanting human tumors into mice. Q: What is the significance of this model?
A: Screen medicine to serve the people. (Please pay tribute to the great mouse!)
Q: Why don't you skip the direct clinical practice to build a tumor model? Can you also screen the medicine?
A: Well, good idea, or do you come first?
.......
Q: What is CDX, PDX?
A: What are you eager to listen to?
As early as the early 1990s, the National Cancer Institute (NCI) was based on nine different types of tumors (brain, colon cancer, leukemia, lung, melanoma, ovarian cancer, kidney cancer, Cancer cell lines of 60 cancer patients with breast cancer and prostate cancer have introduced a "disease-oriented" drug screening strategy. Simply put, human cancer cell lines are first used for high-throughput in vitro drug screening. CDX model for in vivo verification, [Cancer Cell Lines for Drug Discovery and Development]
Since the CDX model cell line is easily available, there are a large number of published literatures on its genomics, cell function and pharmacodynamic response for reference, and the cost of modeling is low. [PDX model and individualized drug guidance] At that time, it can be said that it is popular in major laboratories.
However, in 2016, NCI decided to “retire the NCI60 cell line that has been in use for 25 years from its drug screening system!
Why is this change happening? !
Originally, researchers have gradually found that human tumor cell lines have long-term in vitro culture, and their tumor cell biological behavior, gene expression level, and tumor heterogeneity are significantly different from the original tumor tissue, thus predicting clinical efficacy. The aspect is not ideal. Studies have shown that only about one-third of the drugs identified by this model are validated in Phase II clinical trials [Relationships between drug activity in NCI preclinical in vitro and in vivo models and early clinical trials.] Application and development in translational medicine]
However, after the waves of the Yangtze River push the waves and retire the CDX, there will be a better tumor model to rise! This is the next small series to introduce PDX for everyone~ 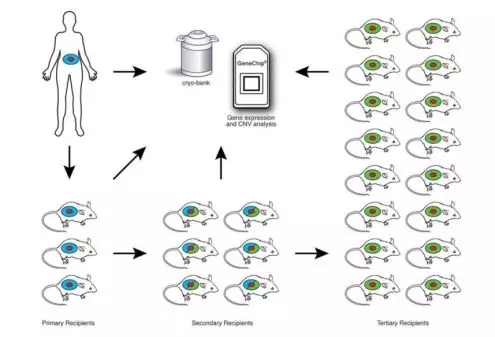
Fig. 1 Engraftment and expansion of patient-derived solid tumor xenografts. Young adult NSG mice are engrafted with solid tumors (shown as red) Human stroma accompanying the tumor is shown in blue. Primary solid tumors are engrafted subcutaneously, orthotopically, or under the Renal capsule. Established grafts are excised from primary recipients, a portion is cryopreserved, and a portion is analyzed for gene expression, including copy number variation (CNV). The remainder is expanded through serial transplantation in secondary and tertiary NSG recipients to generate cohorts of Sufficient size for therapeutic trials. At each stage tumor samples are cryopreserved and gene expression analyzed to compare with the primary tumor analyzed directly ex vivo from the patient. The primary recipients of solid tumors retain human-derived stroma (blue). Tumors in secondary recipients Contain a mixture of human-derived and mouse-derived stroma (mixture of blue and green). Tumors
In tertiary recipients contain predominantly mouse stroma (green) [Human Cancer Growth and Therapy In NOD/SCID/IL2Rγnull (NSG) Mice]
Since the CDX that has been used by the research community for decades to be "retired" will necessarily represent the new force PDX is blue. At present, the advantages that the model is generally recognized by researchers mainly include the advantages and disadvantages of the human tumor PDX transplantation model:
1. The specimens used for transplantation are directly derived from human tumor tissues, and have not been cultured in vitro , and stably retain the genetic characteristics, histology and phenotypic characteristics of the tumor, that is, tumor heterogeneity;
As shown in the figure below, for the constructed human colorectal cancer mouse transplantation model, tissue staining with different morphological features revealed that in some cases, both fresh lesions and passaged lesions showed a good differentiated phenotype, some The samples showed clear cells, mucus secreting cells and necrotic areas. At the same time, a small number of tumors showed high pleomorphism and remained undifferentiated (Fig 2B). 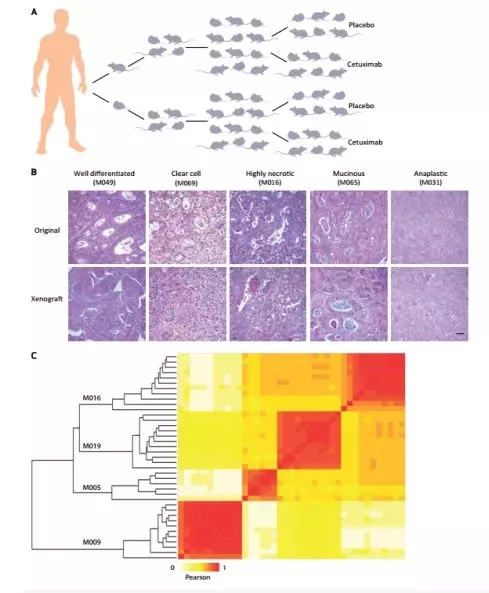
Figure 1. Setup and characterization of the xenopatients. A, generation of xenopatients. After surgical removal from patient, each metastatic colorectal cancer specimen was cut in small pieces and 2 fragments were implanted in 2 mice. After engraftment and tumor mass formation, the Tumors were passaged and expanded for 2 generations until production of 2 cohorts, each consisting of 12 mice. These were randomized for treatment with placebo (6 mice) or cetuximab (6 mice). B, xenografted tumors retained the histopathologic characteristics of original samples. Hematoxylin and eosin stains of representative cases with different morphologic features. In some instances, both fresh and passaged lesions displayed a well-differentiated phenotype, with cells describing irregular pluristratified tubular/acinar structures with minor lumens embedded in a scarce stromal matrix. a clear-cell appearance or featured high nuclear grade and areas of necrosis. In some cases, di Scohesive mucus-secreting cells defined moderately differentiated phenotype typical of mucinuos adenocarcinoma, with signet-ring elements showing peripheral nuclear delocalization and abundant intervening stroma associated with desmoplastic reaction. Finally, a few tumors exhibit high-grade pleomorphism and could be pathologically classified as poorly Differentiated adenocarcinomas. Scale bar, 50 μm. C, genetic concordance between xenografts and their original counterparts. Similar groups of samples are evidenced by applying a Pearson-based hierarchical clustering to copy number calls
2. PDX can be used to screen chemotherapeutic drug sensitive or drug resistance markers, and the test results have good clinical predictability.
Taking gastric cancer PDX as an example, Panel A shows the difference in EGFR DNA/mRNA/EGFR receptor protein copy number in sensitive and sensitive mice treated with cetuximab, and found that the copy number is generally higher in sensitive mice, and through tumor growth curve, IHC Compared with FISH, it was concluded that the expression of EGFR was generally up-regulated in a mouse model sensitive to cetuximab. 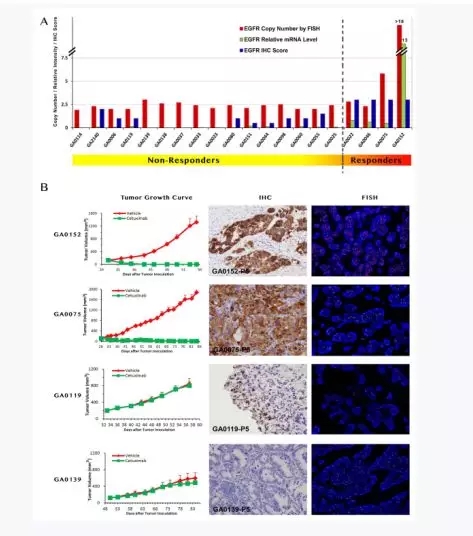
Left: Representative tumor growth curves of responders and non-responders. Middle: IHC analysis of tumor models; Right: Dual-color FISH assay in gastric carcinoma. Probe for EGFR locus is labeled in red and CEP7 labeled in Green. Blue: Nuclei.
In addition, PDX preserves the tumor stroma and stem cell components better during the transplantation process, making the tumor microenvironment closer to the actual situation, and providing a large number of specimens for the preservation and passage of tumor samples.
At the beginning, Xiaobian also said, what is modeling for? Sieve medicine!
These tumor-bearing mice can be used as a patient's "avatar" to replace the patient's treatment effect of different drug regimens, and to screen out the most effective treatment plan to avoid drug side effects and economic waste [PDX model and Tumor individualized medication guidance], the specific process refers to the following figure. 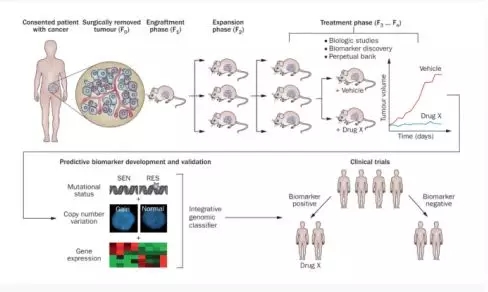
Establishment and testing of PDTX models. Excess tumour specimens not needed for clinical diagnosis are obtained from the consented patients (F0). Non-necrotic areas of these tumours are sectioned into ~3 mm3 pieces and, after processing, implanted subcutaneously into anaesthetized 5- Week to 6-week-old female athymic nude mice. During the engraftment phase, tumours are allowed to establish and grow and then are harvested upon reaching a size of 1,500 mm3 (F1). Similar protocols are employed for subsequent expansion cohort (F2) And treatment cohort (F3 ... Fn). Typically, biological assays are performed on tumours in early generations (≤F5); these biological assays include drug efficacy studies, rational combination studies and the development of predictive biomarkers for novel targeted therapies. Biomarkers Being accurate prediction in a validation set of PDTX models (or 'xenopatients'), they might be translated into early phase clinical trials as tools for patien Ab selections. Abbreviations: PDTX, patient-derived tumour xenografts; RES, resistant; SEN, sensitive. [Patient-derived tumour xenografts as models for oncology drug development]
Of course, although PDX has many advantages, the limitations cannot be ignored [Application and development of PDX model in tumor transformation medicine]
At present, the main source of the original tumor in the PDX model is surgical resection. The modeling is difficult and cannot be repeated. The construction time is long and the success rate is unstable. As the number of passages increases, the tumor microenvironment will gradually be replaced by the mouse extracellular matrix. There are certain restrictions on the number of passages. It is worth mentioning that mice bearing tumors are all immunodeficient mice, so this model can not be used to screen immune-related drugs.
In general, the two models have their own advantages. To some extent, CDX can be considered as a PDX model with too many passages and it is difficult to trace. [PDX model and individualized drug guidance] After alternate passage in vivo and in vitro, in the original tumor tissue. Only individual clones that are most suitable for in vitro culture conditions are retained, and clonal heterogeneity is lost, making it difficult to predict clinical efficacy. The biggest advantage of PDX is also here, which preserves tumor heterogeneity and is more in line with clinical tumor characteristics.
The company's headquarters is located in Yizhuang, Beijing. It has branches in Shanghai Zhangjiang, Jiangsu Haimen, Boston, USA, and also in Wuhan, Guangzhou, Chengdu and other places. There is an office.
Established in the United States in 2008 and successfully operated. In 2009, Beijing Baiao Saitu Headquarters was established, and in 2012, the headquarters was relocated to Beijing Yizhuang Economic and Technological Development Zone.
Bio-Saitu is a high-tech company based on a stable and efficient gene editing technology platform, with model animal customized services, important model animal development and large-scale breeding and supply, in vivo pharmacological efficacy evaluation services, and antibody drug research and development outsourcing services. Biopharmaceutical companies.
The company adheres to the principle of pursuing better human health and aims to provide high quality products and services for new drug research and development.